Abstract
INTRODUCTION. BCL2 homology 3 (BH3) mimetics represent a novel class of pro-apoptotic anti Hematologic malignancies (HM) drugs directly targeting apoptosis, even in chemoresistant cells. Each disease is heterogenous which makes it hard to provide standardized treatment for each patient and highlight the need for new diagnosis tools to deliver precision medicine. In the other hand, a technic called BH3 profiling has been developed, allowing to measure the apoptosis priming of the cells as well as their dependencies on specific anti-apoptotic proteins. Using specific BH3 peptides, this assay measures the percentage of cytochrome c release for each anti-apoptotic protein, mainly Bcl2, Mcl1 and Bcl-xl. BH3 toolkit, a variation of this technic, was developed using BH3 mimetics instead of BH3 peptides. Those small molecules mimic the effect of BH3 sensitizer protein by competing for anti-apoptotic protein catalytic pocket. Using these drugs allows to avoid cell permeabilization and thus reduce variability in the assay. A dynamic BH3 toolkit is also possible, measuring early drug-induced changes in net proapoptotic signaling, to identify modification of antiapoptotic dependencies.
The aim of the present work is to develop a standardize and reproducible BH3 toolkit (baseline and dynamic), with internal control, to allow an absolute measurement of specific anti-apoptotic dependencies in HM cell lines or primary tumor cells, as well as identifying optimal therapeutic combination.
MATERIAL AND METHODS. We used different HM cell lines as well as primary cells from patients with CLL. Cells were seeded right after thawing at the concentration of 200,000 cells/mL in a 96 well plate, using a robotic liquid handler. Primary cells were cocultured with stromal cells (Resto-6 cell line). Cells are then incubated for 20h in a 5% CO2 and 37°C incubator. Next, we treated them with BH3 mimetics (or vehicle): Venetoclax (VEN, Bcl2 inhibitor), Navitoclax (Bcl2 and Bcl-xl inhibitor), AZD-5991 (AZD, Mcl1 inhibitor), A-1155463 (Bcl-xl inhibitor) for 4h. Then, cells were stained using Annexin V and 7AAD and analyzed by flow cytometry. Our gating strategy focuses on live cells to measure annexin V flipflop as an apoptotic marker. This assay is paired with a cell viability measurement at 24h, using CellTiterGlo assay. For dynamic BH3 toolkit, the influence of the tested drug on antiapoptotic dependencies is measured by adding this drug (or vehicle) at the same time as cells of interest.
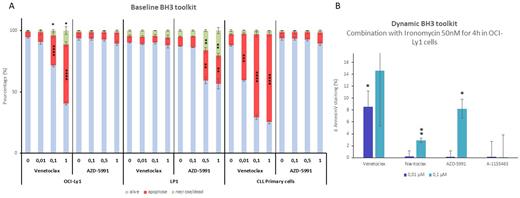
RESULTS. First, we wanted to optimize the protocol on cell lines with known anti-apoptotic dependencies. We used OCI-Ly1 and LP1 cell lines which are known for their exclusive Bcl2 and Mcl1 dependencies, respectively. As expected, OCI-Ly1 cells showed a significant increase in annexin V staining (reaching 48.2% (P=0.0003) at maximum concentration) upon VEN treatment, whereas it does not change significantly with Mcl1 or Bcl-xl inhibitors (Figure A). Likewise, we detected an increase of 15.35% (P=0.005) in Annexin V staining only upon AZD in LP1 cells, but no significant modification with other inhibitors. Consistent with these data, we saw a significant and specific decrease in cell viability (CTG) with VEN and AZD on OCI-Ly1 and LP1 respectively (data no shown). These 2 cell lines are used as internal controls for the other analyses.
Next, we optimized the assay on CLL primary cells. Figure A shows that VEN induces an increase in Annexin V staining by 62.7 % (P=0.0003), demonstrating a significant Bcl2 dependency.
Finally, we optimized the dynamic part of the assay (Figure B). We used ironomycin, a lysosomal iron-targeting small molecule that has been studied by our team in HM (Devin et al. 2022). We found that ironomycin increased significantly Bcl2 dependency in OCI-Ly1 cells by 8.55% (P=0.02) with VEN. Interestingly, we observed the induction of a MCL1 dependency in those cells, with an annexin V staining increase by 8.21% (P=0.02).
CONCLUSION. We were therefore able to measure the baseline dependencies using a BH3 toolkit that avoid cell permeabilization. We were also able to measure changes in dependencies induced by an innovative therapy. We believe that our results show the robustness of the BH3 toolkit and its ability to detect potential new therapeutic strategies in HM cell lines and primary cells. Detailed results will be presented at the meeting, as well as data on primary lymphoma and myeloma cells.
Disclosures
Herbaux:Takeda: Honoraria, Research Funding; Gilead: Honoraria; Janssen: Honoraria; Roche: Honoraria; MSD: Research Funding; Abbvie: Honoraria, Research Funding; Kite: Honoraria.
Author notes
Asterisk with author names denotes non-ASH members.

This feature is available to Subscribers Only
Sign In or Create an Account Close Modal